Ensuring Mold Steel Polishability
When working on a project that requires high-quality texturing or surface finish, using an electro-slag quality material may be the best option for mold steel.
When it comes to surface finish, it must be understood that any defect that appears on the surface of the mold will be replicated onto the surface of the molded part. If the mold surface contains scratches, pits, welds, uneven textured patterns, etc., and they are visible on the mold surface, they most likely will appear to some degree on the molded part. In some cases the molded component may not have any cosmetic or aesthetic requirements. If so, some surface flaws on the part may be acceptable and not considered to be a cause for rejection.
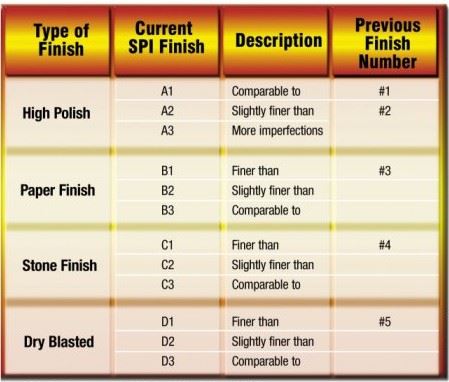
In other circumstances, the finished part may need to function as a lens or may have a consumer application that requires a smooth, flawless appearance. Under these conditions a nearly perfect, polished mold surface will be necessary. In an effort to define such a subjective parameter as mold material polishability, a scale defined by the Society of the Plastics Industry (SPI) was created. If the surface finish of the molded part will be a high priority, one must consider the use of a mold material that will reach what is commonly referred to as a number one, or more specifically an A1 finish (see Figure 1).
The producers of mold steels will often utilize a process known as electro-slag remelting (ESR) to produce materials that are capable of being polished to a high surface finish. This additional step in the steel manufacturing process helps ensure that the mold steel will contain a low-level of non-metallic inclusions. Non-metallic inclusions are brittle constituents, such as oxides and sulfides that are formed within the mold steel due to either the presence of tramp elements or the influence of the atmosphere during the mold steel melting and casting process.
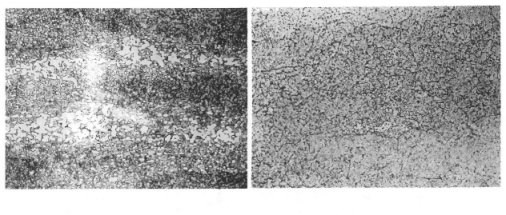
An ESR furnace is basically a movable copper mold that contains a basic slag. The heat of the slag is used to melt the as-cast ingot. The ingot melts droplet-by-droplet and the dense steel falls through the slag and re-solidifies at the base of the mold. The slag acts as a filter, absorbing sulfur. In addition, the relatively fast re-solidification results in a material with a relatively low level of segregation. The result is a steel with a low inclusion level and a homogeneous microstructure (see Figure 2).
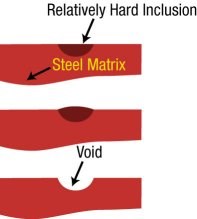
How does the presence of non-metallic inclusions and segregation within the mold steel limit the integrity of a mold steel? Let's refer to Figure 3, which schematically represents the presence of a sulfide inclusion contained within a mold steel.
After the grinding process, any such inclusions will be nearly undetectable. However, during the stoning/polishing operation, the softer steel matrix that surrounds the hard, brittle sulfide will be preferentially removed. Effectively, the inclusion is "lifted" from the surface. As the polishing operation continues, the inclusion is pulled from the matrix of the steel and a void or pit is left behind on the steel surface. An actual example of a pitted surface is shown in Figure 4.
Polished Performance
Since polishing is typically the last operation performed prior to putting the mold into production, such a problem is not detected until the final stages of mold construction. Once such defects are found, one must stone and re-polish, in hopes of finding a plane within the steel that does not contain these inclusions. To avoid these issues, it is imperative that when surface appearance is a high priority you select an electro-slag remelted (ESR) mold steel.
Polishability of mold steel also is a function of its chemical composition and its hardness level. By definition, mold steel is comprised predominantly of iron with typically no more than a two percent addition of carbon. In essence, the mold steel - or any alloy for that matter - is a solution, or more specifically a solid solution made up of iron, carbon and any number of alloying elements. Some common alloying elements include chromium, molybdenum and vanadium. These elements may be defined as carbide-formers, since they react with the carbon to form a combination of chromium, molybdenum and/or vanadium carbides. The carbides that are present within the matrix provide the steel with its wear resistance. The greater the amount of carbon and alloy elements, the greater will be the percentage of wear-resistant carbide particles that will form within the material.
However, a problem can develop when these carbides become segregated and form as a banded structure within the material. To provide an understanding of this phenomenon, one needs to consider the microstructure of the mold steel. The microstructure, as the name implies, is an examination of the material under high magnification. Basically, a sample of the material is polished to a mirror finish, etched with an acidic solution and examined with a metallurgical microscope. An example of some mold steel microstructures is shown in Figure 5.
The carbides that form within the mold steel are relatively hard particles held within in a softer, more ductile matrix. The more carbides present, the greater the wear resistance of the steel. However, under some circumstances their presence may reduce the material's toughness or crack resistance. The carbides are relatively brittle and when they form as large, blocky particles they can act as initiation sites for cracking and crack propagation.
This situation will not only be detrimental to the material's crack resistance, but it also may affect the mold steel's polishability. As previously mentioned, the segregated carbide bands are harder than the surrounding matrix. When the polishing operation occurs, the surface of the steel is abraded away by a series of finer and finer scratches. As the polisher works the surface of the steel with a stone, abrasive paper, etc., the hard carbide particles will resist this abrasion relative to the softer surrounding steel matrix. On a microscopic level this will create a series of peaks and valleys that, when viewed with the naked eye, appear as "wavy-ness" on the mold surface.
In addition, if a texturing operation is required, the etchant used to create the textured pattern will react differently across the mold's surface. This is due to the fact that the segregated or banded regions have a different chemical composition. The result is that the banded or segregated areas will etch differently from the surrounding matrix. This difference may appear as "streaking" or a non-uniform depth-of-texture.
Problems with polishing and texturing will present themselves from time-to-time. Unfortunately, such issues occur during the final stages of the mold building process, when delays or re-working become costly. For these reasons one should consider the use of an electro-slag (ESR) quality material. When surface finish or texturing is a high priority, choosing an ESR-quality mold steel will help ensure that you are using the optimum material.
Related Content
It Starts With the Part: A Plastic Part Checklist Ensures Good Mold Design
All successful mold build projects start with examining the part to be molded to ensure it is moldable and will meet the customers' production objectives.
Read MoreMaintaining a Wire EDM Machine
To achieve the ultimate capability and level of productivity from your wire EDM on a consistent, repeatable and reliable basis, regular maintenance is a required task.
Read MoreHow to Analyze and Optimize Cutting Conditions to Reduce Cycle Time
Plastic injection mold design and manufacturing company puts NC program optimization software module to the test. The results were surprising.
Read MoreMold Design Review: The Complete Checklist
Gerardo (Jerry) Miranda III, former global tooling manager for Oakley sunglasses, reshares his complete mold design checklist, an essential part of the product time and cost-to-market process.
Read MoreRead Next
How to Use Continuing Education to Remain Competitive in Moldmaking
Continued training helps moldmakers make tooling decisions and properly use the latest cutting tool to efficiently machine high-quality molds.
Read MoreOvercoming Pain Points in Moldmaking with AI
Shops that embrace AI as a tool, not a threat, can enhance efficiency, preserve expertise, and attract tech-savvy talent.
Read MoreYour Guide to Smarter, Faster Mold Design
Dive into expert-curated content delivering proven solutions for mold optimization, manufacturability and precision performance.
Read More