Tool Steel and Heat Treatment, Part 2
Tool steel, its classification, heat treatment and how to consider manufacturing methods in relation to heat treatment and the metallurgy of heat treatment.
In order for tool steels to be identified by a toolmaker, it is necessary for the toolmaker to understand the method of identification and classification of tool steels. The purpose of gaining this understanding is that there are so many different tool steel manufacturing companies that will produce a very high grade of tool steel and name it a particular brand.
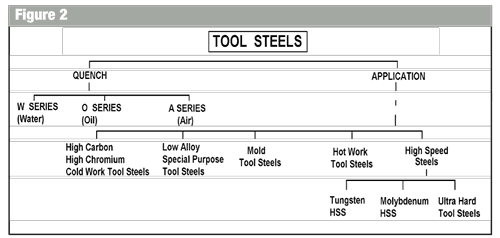
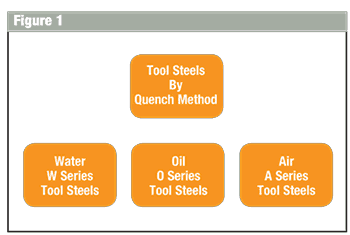
The American Iron and Steel Institute devised a very easy system of identification. The method of identification considers at least nine different types of tool steel quench or application. The first two groups are liquid quench and air. Figure 1 illustrates these groups. Figure 2 shows the ‘tree’ that connects the tool steels together by both quench and industrial application.
Applications
The application steels are characterized by the AISI as follows:
| High carbon/high chromium |
D series (Dimensionally stable steels) High hardenability, secondary hardening steels (2% carbon, 5% chrome). |
| Low alloy special purpose tool | L series. Generally a water quench steels. |
| Mold tool steels | P series. Usually low carbon steels, High chrome with moly and nickel. |
| Hot work tool steels | Chromium Hot Work (0.40% carbon, 5% Chrome (H10 TO H19) |
| Hot work tool steels | Hot Work (0.35% carbon, 3% to 5% chrome, 10% to 15% tungsten) (H21 to H26) |
| Hot work tool steels Hot work steels |
Molybdenum Hot work (0.60% carbon) 4% chrome, 1.0% Vanadium, 15% Tungsten, 5% Moly (H42) |
| Tungsten high-speed steel | 0.80% carbon (nominal), 4% chrome 16.5% (nominal) tungsten, 1% Vanadium T1 to T15 |
| Molybdenum high-speed steel |
M series HSS M1 to M36 1% carbon (nominal), 4% chrome, 2% vanadium (nominal) 2% tungsten (nominal) 7% molybdenum (nominal) Some steels have up to 12% Cobalt |
| Ultra hard high-speed steel |
M series M41 to M47 1.10% carbon (nominal) 4.0% chrome(nominal) 3.25% tungsten (nominal) 7.5% molybdenum (nominal) 7.75% cobalt (nominal) |
All of the above nominal values will change according to the numeration of the tool steel.
Characteristics
Water Hardening Tool Steels: Low hardenability, water quench necessary to form martensite.
Oil Hardening Tool Steels: High wear resistant steels.
Air Hardening Cold Work Tool Steels: High wear resistance. Requires the surface to be protected against surface oxidation and decarburization. Because of the high carbon content, fine carbide distribution is accomplished. Will form martensite with an air cool; however, scale and decarburization will occur. Therefore it is necessary to process under vacuum conditions or protective atmosphere. Because of the slow cool requirement to form martensite, distortion is kept at a minimum, except for induced residual stress resulting from prior machining.
High Carbon/High Chrome D series: These steels are known as secondary hardening steels. Some of the alloy carbides will form on the tempering of the steel. In other words, the steel will increase in hardness value to form large volumes of the alloy carbides, giving high wear characteristics. It should be noted that these steels (although known as dimensionally stable steels) will distort if the induced machining stress is not relieved before the austenitizing process. They are typically used in cold work applications.
Mold Steels (P series): Because of the low carbon content, the hardenability of the steel is low. These steels will be suitable for hobbing of cavities for plastic mold dies or low-temperature metal casting. The only method of achieving any degree of high surface hardness for wear resistance is to surface treat by FNC, carbo-nitride or carburize.
Chromium Hot Work Die Steels: This group of steel is generally used in hot work applications such as forging, aluminum extrusion, shot sleeves, magnesium die-casting and aluminum die-casting. These steels have good hardenability and will harden (as quenched to approximately 55HRC depending on the maximum cross sectional thickness).
Tempering temperatures are generally in the 1000°F range so that the carbide precipitation will begin to occur (vanadium and chromium carbides). This steel group is also susceptible to retained austenite, which can and must be decomposed by multiple tempering.
Molybdenum Hot Work Tool Steels: These steels are employed when higher forging temperatures are being used, as opposed to temperatures used on alloy carbon steels. This group of steels is used for high temperature forging, hot extrusion or hot metal die casting—such as aluminum die casting and magnesium die casting.
Molybdenum High-Speed Steels: Molybdenum is among the carbide forming elements and strongly assists in the formation of hard carbides that are finely dispersed throughout the matrix of the steel. Care must be taken with these steels with the austenitizing temperature selection as well as the holding time at temperature. Eutectic grain boundary melting can begin to occur if the steel is held too long at too high an austenitizing temperature.
Tungsten High-Speed Steels: Tungsten is a very strong carbide former and will produce very high as quenched hardness values. All of the high-speed steels (molybdenum and tungsten) are known as secondary hardening steels. In addition, this group of steels has the ability to cut at red heat without losing its hardness value. The addition of cobalt enables an even higher operating temperature, before losing the as quenched hardness.
Ultra Hard High-Speed Steels: These steels are used to produce hardness values in excess of 70 HRC. The ultra hard high-speed steels will demonstrate high impact values. In other words, they are very tough under repeated impact such as milling cutters.
Carbide Formers: Carbide formers are those elements that will form strong carbides of finely dispersed carbides. The strong carbide forming elements are chromium, molybdenum, tungsten, vanadium, manganese, titanium, zirconium and niobium.
The above elements will react favorably with the carbon to form hard carbides, provided that the appropriate austenitizing temperature was selected. It is not necessary to have all of the above elements in solution. Provided there is carbon (in solution) then these elements can be present individually or corporately. The non carbide forming elements are nickel, aluminum, copper, nitrogen, cobalt and silicon.
Terminology
Grain Size: All metals have grains (just as salt is in a granular form; but some grains can be larger than others). The size of the grain in steel is influenced by temperature, time at temperature and steel analysis (composition).
Hardenability: Hardenability is the ability of the steel to produce the appropriate hardness in relation to the amount of martensite when quenched for its austenitizing temperature into the appropriate quench medium. The resulting hardness will be according to its cross-sectional area and chemistry (composition).
The hardenability test is measured by heating a test piece of a very specific size, followed by heating the test bar and quenching the end face. This test is known as the Jominy End Quench Hardenability End test.
Hardness: Hardness is the measurement of the steel’s resistance to indentation by pressing into the surface of the steel a specific object—such as a steel or molybdenum ball or diamond indenter—using very specific loads. Hardness is simply the resistance to indentation as determined by load over area of the specific indenter.
Heat Treatment: Heat treatment is any method of applying heat to a metal. The source of heat can be gas, oxy-acetylene, electricity micro-wave, laser, etc. There are always three aspects to the heat treatment cycle: (1) heat up to the selected temperature, (2) soak at that temperature and (3) cool down from that temperature.
Holding Temperature: The holding temperature is that temperature that has been selected for the specific process underway. The term always refers to time at temperature, meaning the time that the steel reaches the selected process temperature, not when the furnace reaches temperature.
Hypo-Eutectoid Steel: Hypo-eutectoid steel has a carbon content of less than 0.77% present in solution.
Induction Hardening: Induction hardening is a method of steel surface heat treatment. In order to be able to induction heat treat (harden) the surface of a steel, there are three prerequisites necessary: (1) sufficient carbon in the steel to transform to a martensitic structure at the surface of the steel; (2) appropriate austenitizing (hardening) temperature for the steel surface to transform to austenite; (3) appropriate cooling rate that will transform the created and formed austenite into martensite at the steel surface.
Induction Heating: Induction heating is a method of heating a metal by electrical energy that will create an electrical resistance and hysteresis, generating heat at the steel surface by creating rapid electro magnetic energy at the surface. However, induction is not only used for hardening. Other processes that can be accomplished by induction technology include induction hardening, tempering, heating of steel billets for forging, heating for forming and brazing.
Intergranular Cracking: Intergranular cracking is a condition in the steel that that will occur between the grain boundaries of the steel. Generally (but not in all cases), it is caused by the diffusion of oxygen into the steel surface that leads to a condition known as intergranular oxidation (IGO). IGO will initiate fractures at the granular faces and grain boundaries. It is caused by heating in an oxygen-rich atmosphere or prolonged soak times at elevated carburizing temperatures.
Isothermal Transformation Temperature: The isothermal transformation temperature is commonly known as the Triple T Diagram (TTT). It is a diagram that is used to assist the heat treater in knowing how to select the critical cooling curve of the steel being quenched or cooled to achieve a particular metallurgical structure. It also is known as the isothermal transformation diagram (IT diagram). This diagram is consulted for a process known as isothermal annealing.
Ion Nitriding: Ion nitriding is a surface treatment method of diffusing nitrogen in the surface of the steel to enable the nitrogen to react with alloying elements that will readily form stable nitrides. This will result in a hard surface layer in the immediate surface of the steel. The appropriate gases are ionized electrically in a specially-designed, low-pressure vacuum process chamber. The process is also known as plasma nitriding, glow discharge nitriding and ion nitriding.
Ion Carburizing: This surface treatment process generates carbon for diffusion into the steel surface. Once the carbon has been diffused to the appropriate depth, the treated steel is austenitized according to the surface carbon content and cooled at the appropriate rate to transform the induced carbon/austenite into martensite. In effect, the process is very much like gas carburizing with much the same results. It is a surface treatment. The process is accomplished under similar electrically generated conditions as with ion nitriding—with the exception of the higher process temperature.
Intensive Quenching
This is a patented process using water as the primary quench medium to transform the created austenite into martensite. The process is reported to be successful with highly alloyed tool steels, including high-speed steels, the D series and the H series.
Summary
Just as one designs, engineers, calculates and manufactures a mold, the same applies to the design of tool steel, and if tool steel is to perform well there needs to be a solid combination of chemistry, steel making, machining and heat treatment.
Related Content
Modified Composition Enhances Stainless Steel Capabilities
Edro introduces Böhler’s M333 Isoplast stainless steel which has been manufactured via a new melting technology for best-in-class polishability, conductivity, toughness and corrosion resistance.
Read MoreHow to Produce More Accurate Molds and Reduce Rework
Patented micro-milling process for manufacturing steel plate flat and parallel helps mold builders shorten mold build times and increase accuracy.
Read MoreMoldMaking Technology's Top-Viewed Content in 2022 So Far: Features
A list of the most-viewed features in MoldMaking Technology from January-May 2022, from vertical machining to achieving the best mold finish. Give your favorites another look or catch up on what you might have missed.
Read MoreMoldmakers Deserve a Total Production Solution
Stability, spindle speed and software are essential consideration for your moldmaking machine tool.
Read MoreRead Next
Tool Steel and Heat Treatment, Part 1
An introduction to heat treatment for the moldmaker.
Read MoreAre You a Moldmaker Considering 3D Printing? Consider the 3D Printing Workshop at NPE2024
Presentations will cover 3D printing for mold tooling, material innovation, product development, bridge production and full-scale, high-volume additive manufacturing.
Read MoreHow to Use Strategic Planning Tools, Data to Manage the Human Side of Business
Q&A with Marion Wells, MMT EAB member and founder of Human Asset Management.
Read More







.jpg;maxWidth=300;quality=90)