Applying cBN Technology to Moldmaking
A summary of cBN and its progression to a preferred substrate for high-accuracy hard milling.
The advent of new technology frequently changes the manner in which ideas are transformed into tangible realities. The synthesis of hexaganol boron nitride, (h) BN, to cubic boron nitride, cBN has positive implications with respect to how materials in the die/mold industry are transformed into finished products. To properly review the fundamental advantages of cBN technology, it is important to first understand some background information describing what it is and how it is made. Once a basic foundation has been established, a more detailed analysis will be given with respect to unique properties of cBN, its various forms and practical applications of each. Lastly, further explanation and examples will be discussed in reference to specific cBN applications in the die/mold industry. Since the implications of cubic boron nitride are similar to those polycrystalline diamond (PCD) technology had on milling just a few years ago, it is important to understand the fundamental characteristics of cBN and how it can be applied to benefit our industry.
Background
Boron nitride was discovered in the nineteenth century. The first synthesis was in 1842 by W.H. Balmain, who used the reaction of molten boric acid on potassium cyanide to create it. Unfortunately, the resultant material was extremely unstable. It wasn’t until the 1950s that more stable versions of boron nitride were developed in the shape of powders and hot-pressed shapes. However, the lack of technology in the early 1950s made the synthesis of h-BN and c-BN very unpractical for commercial purposes due to the extreme cost of production. Only in the last 15 years—with the advent of newer technology—has the commercial production of boron nitride been economically advantageous.
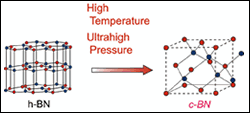
Perhaps the best way to understand the physical properties of boron and nitrogen is to compare them to another similar element that is already well known: carbon (or graphite). Boron and nitrogen have many similarities to that of carbon in terms of composition. For example, boron and nitrogen combined have the same number of outer electrons as carbon. Moreover, the atomic radii of boron and nitrogen also are very similar to that of carbon. These similarities are not all that shocking, as boron and nitrogen are adjacent to carbon on the periodic table. Thus it should be no surprise that boron nitride and carbon also share very similar crystal structures. Just as carbon exists as both graphite (hexaganol) and diamond (cubic), boron nitride exists in the same two forms. The synthesis of both elements from the hexaganol form to the cubic form is done through the application of high pressure at high temperatures.
Polycrystalline cubic boron nitride is a composite formed by sintering together cBN particles in the presence of a metal or ceramic catalyst binder material. The synthesis of hBN to cBN occurs at pressures in excess of 80,000 atm and temperatures over 2000oC. In comparison, diamond is synthesized from graphite in a similar fashion, but the pressure required to change the crystal structure of graphite to diamond is approximately 5000 atm (17x less than that to synthesize c-BN from h-BN). The vast amount of pressure and heat required to create cubic boron nitride from h-BN is the main reason why it is so expensive. Research to synthesize cBN at lower temperatures and pressures is ongoing (see Figure 1).
Properties
Polycrystalline cBN shares many of the same qualities that PCD possesses. Diamond has high thermal conductivity, excellent chemical resistance, and is ideal for machining in non-ferrous materials. CBN also has excellent thermal conductivity, is chemically inert, and at 4500Hv is second in hardness only to diamond. However, these characteristics are where the similarities stop.
While diamond is ideal for use in non-ferrous (materials with no iron content) applications, it is not suited for machining in ferrous materials. The reason it cannot be used is because diamond essentially dissolves in iron. The heat generated through friction in a ferrous material in the presence of air causes the diamond structure to decompose into carbon dioxide. For this reason, diamond cannot be used to cut steel.
This is where the fundamental difference between diamond and cBN exists. Cubic boron nitride has virtually no reactivity with iron. It is stable in air up to 1400oC, whereas diamond decomposes above 800oC. Cubic boron nitride has increased toughness over diamond, and with the onset of new production technology, now offers cost-effective performance in cutting tool applications in steel up to 68 Rockwell-C.
The variations of cBN that can be made by changing certain properties are virtually endless. However, there are four basic properties or components that affect the mechanical properties of polycrystalline cubic boron nitride. They are cBN content, cBN grain size, binder type and format. (See Table 1)
PCBN in the Die/Mold Industry
The majority of the die/mold industry uses two types of tools for 3-D machining: a titanium aluminum nitride (TiAlN) coated carbide tool for applications in die steels and a CVD (Chemical Vapor Deposition) diamond coated tool for applications in graphite.
|
Table 1
|
||||
|
cBN Content
|
cBN Grain Size (µm)
|
Binder
|
Format
|
|
| Type 1 |
90%
|
8
|
Al
|
Solid
|
| Type 2 |
80%
|
6
|
Ti/Al
|
WC backed
|
| Type 3 |
50%
|
2
|
TiC
|
WC backed
|
| • Type 1: Is an example of a coarse grain grade of cBN. This form—with its high percentage of cBN—possesses high thermal conductivity and has strong implications for grinding operations in hardened steels. The aluminum ceramic-based binder adds additional thermal advantages making it better suited for the high-heat environment typical in grinding. • Type 2: Is a medium grain form of cBN with a tungsten carbide backing that permits more flexibility in terms of tool design. This type of cBN would typically be used as the optimum material for cBN inserts. • Type 3: Is a fine grain cBN with titanium carbide (TiC) based binder material and 50 percent cBN content. This combination is ideal for use in conjunction with smaller cutting tools, where finish and wear resistance in hardened steels are more critical. The lower percentage of cBN particles makes this grade less susceptible to fracture, hence better for light inter-rupted cuts. Furthermore, cutting edges are more stable, enabling superior finishes and longer tool life. (See Figure 2.) Table courtesy of OSG Tap and Die, Inc. |
||||
Any mold builder would agree that if the diamond tooling exemplified the same cost-savings in steel as it did in graphite, diamond tooling would be the only tooling of choice. As previously stated, diamond cannot be used in applications with ferrous materials, hence PVD (physical vapor deposition) coated carbide end mills are used in conjunction with die steels. The downside of PVD coated carbide tools is evident when milling in materials above 55 Rockwell-C. Stringent geometric tolerances and demands on surface finish make it difficult for some mold builders to machine hardened steels in a manner that is cost-effective. A hardened block of D2 (60HRC) can wear out a four-millimeter ball end mill in less than one hour under the most conservative milling conditions.
Unfortunately, the finishing stage of milling is the longest cycle in the machining process, and can easily consume three or four tools in the finishing stage alone to complete one part. Rapid and uneven flank wear on the cutting edge leads to multiple tool changes—making it extremely difficult to maintain tight geometric tolerances while simultaneously achieving mirror finishes. The end result is added time for benching and polishing—both of which are tertiary operations that directly reduce profit margins. This is where the fundamental advantage of cBN technology proves invaluable.
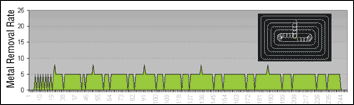
When ground cutting tool geometry is applied to a cBN substrate similar to that of Type 3, tool life in a finishing application can exceed 10 times that of the same tool from a carbide substrate. Excellent cutting edge retention is experienced due to the high hardness and toughness of the TiC based cubic boron nitride. In addition to supreme wear resistance, cBN-cutting tools also can be run significantly faster than that of carbide. A one-millimeter cBN ball end mill can be run in excess of 40,000 rpms in hardened CPM-4V, 62 HrC. Such a dramatic increase in tool life and speed has obvious benefits. During finishing operations, one cBN tool can be used in place of multiple carbide tools, thus eliminating tool changes and reducing overall tooling costs. No tool changes equal faster cycles times and no tool-change lines on the part. The dramatic increase in spindle speed further optimizes metal removal rates and lowers finishing cut time.
While cBN has tremendous cost-saving implications in the die/mold industry, machining with such a substrate has its limitations. Ground cBN cutting tools are typically for finishing applications only. The brittle nature of the substrate makes it extremely sensitive to radial forces. For this reason, cBN tools are typically reserved for finishing applications where the lighter depths-of-cut equate to less radial load at the tip of the tool. Furthermore, careful attention must be paid to programming toolpaths. Abrupt radial movements (particularly in corners where larger amounts of material exist) can cause breakage. The best strategy for applying cBN technology is often in applications where machining toolpaths have been proven. The utilization of cBN cutting tools for roughing and semi-finishing operations is only feasible with ground, spiral gash point geometry. Such geometry permits more stability by reducing radial cutting forces. At the spindle, the end result is a freer machining cutting tool capable of taking deeper axial depths-of-cut than that of previous ball mills with spade-like geometry. (See Figure 3 and Figure 4).
Summary
Cubic boron nitride technology has come a long way since its original synthe-sis in 1842. Advancements in modern technology have made the commercial produc-tion of cBN not just possible, but economically advantageous. While the price of cBN tools appears to be quite lofty, the proper application of such technology can yield significant cost-savings with respect to both tooling and production costs. With advanced machine tool technology and proven toolpaths, the following can be achieved:
- Reduced cycles times through in-creased spindle speeds.
- Reduced tooling costs through in-creased tool life (10x life of carbide).
- Elimination of benching via 5-micron radius tolerances.
- Elimination of polishing via a sus-tained cutting edge and the elimination of tool-change lines, which combined create mirror finishes.
The positive implications of cBN technology are not unlike those diamond technology had on the die/mold industry during its initial introduction. With this in mind, it is important to view the fundamental application of cubic boron nitride as not just an idea, but as a necessity for future growth in a fiercely competitive industry.
Read Next
Constant Material Removal: The Key To Hard Milling
Features of the CAM software can keep the cut from exceeding the tool’s limits, without compromising part quality or productivity.
Read MoreHow to Use Strategic Planning Tools, Data to Manage the Human Side of Business
Q&A with Marion Wells, MMT EAB member and founder of Human Asset Management.
Read More
















.jpg;maxWidth=300;quality=90)



_300x250 4.png;maxWidth=300;quality=90)