Solving the Part-Sticking Challenge
Low-surface-energy mold coatings can eliminate part sticking, reduce cycle time and increase mold life.
Mold wear and part sticking can be a constant battle and potential profit-killer when it comes to aggressive polymer molding materials. In many cases, master batches (a concentrated mixture of additives) formulated for release can be blended into certain resins to ease part release, but with glass-filled materials wear is still a major issue. Also, molds and cores can be hardened to a level that minimizes wear, but the elevated temperatures cause extreme brittleness. A more viable option is to use a coating to protect the mold and its components while eliminating the part-sticking challenge.
Finding the right coating and then the ideal balance of coating surface energy, hardness, toughness and corrosion resistance that is required for the molding of sticky and/or highly abrasive molding materials is a challenge. Many articles review coatings that appear to meet the required criteria but rarely meet production expectations. Coatings do not come with a “one size fits most” tag attached, but require a cooperative effort between coating providers and customers to determine the best fit for each application.
For example, a company that produces swimming pool accessories was injection-molding polyvinyl chloride (PVC) components and experiencing problems with parts sticking in the mold. The operator had to apply mold release every two shots, which had a major impact on achieving the required cycle times. By applying a nanocomposite diamond-like carbon (DLC) coating, the company was able to reduce the need for mold release to once every 7,000 shots. And the likelihood of mold damage due to digs and scratches from tools used in part removal was eliminated.
The ABCs of DLC
Nanocomposite DLC coatings are basically diamond-like carbon that has been improved through the addition of molecular additives to reduce internal film stress and modify surface properties. These coatings can be applied to cavities, cores and ejector pins made from most alloys used in plastic molding and can increase mold life, eliminate part sticking and reduce cycle times. This is due to a balanced combination of surface energy, hardness, toughness, corrosion resistance, thermal properties, thickness and temperature to ensure maximum performance and durability.
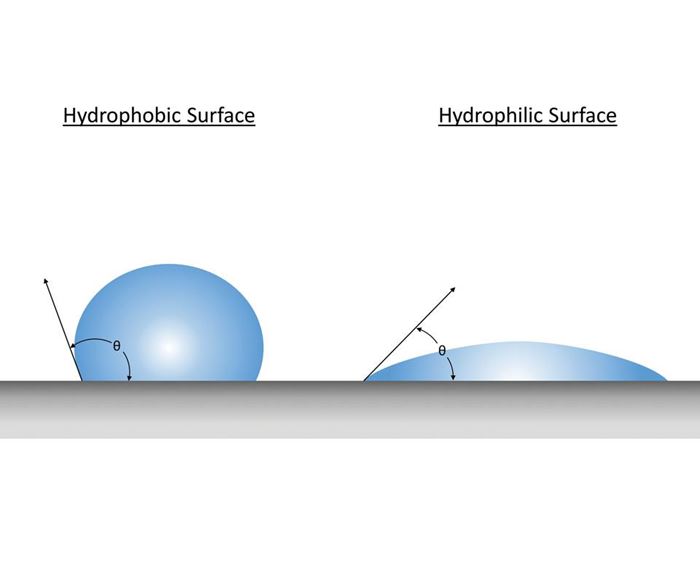
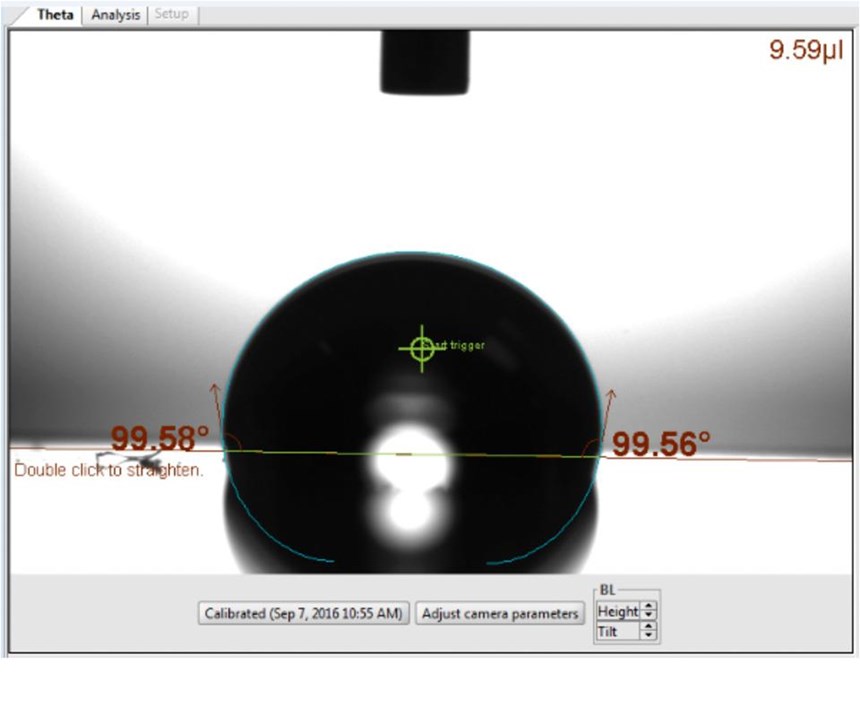
Surface energy. Water contact angle is a common measurement used to determine the surface energy of a material. The surface energy is the energy excess relative to the bulk necessary to form a surface in a crystal. Generally, if the water contact angle is less than 90 degrees, the surface is considered hydrophilic, the tendency for the water to wet or spread on a surface and maximize contact area. If the water contact angle is greater than 90 degrees, the solid surface is considered hydrophobic, the tendency for water to bead up and be repelled, thereby minimizing contact area. DLC coatings have water contact angles as great as 110 degrees, permitting low surface energy that results in reduced bond sites in which polymers can stick to the mold surface.
Hardness. DLCs can produce extraordinarily high hardness relative to the substrate materials. Hardness is the ability of a material to resist plastic deformation when exposed to a compressive force. Often the hardness is an over-emphasized material parameter when considering wear and efficiency applications. Hardness should always be considered in context to the substrate material and the toughness properties of the DLC.
Toughness. The toughness of a DLC can be tailored to the substrate material and application environment. Toughness is the ability of the coating to absorb energy and plastically deform without fracturing. This ideally happens without deformation to the substrate materials.
Corrosion resistance. Corrosion is a diffusive process (the intermingling of particles of two or more substances as a result of a thermal motion), which relies on exposure of the surface to sources of oxygen (most typically) or sulfur. DLC corrosion resistance is rooted in the fact that the refined metal substrates are masked from gradual reduction into their more stable forms, typically by electrochemical oxidation. Basically, the DLC blocks access to reaction sites via the nanocomposite thin film structure being bonded to the surface.
Thermal transfer. High thermal conductivity means better control of mold and core temperatures. For example, when comparing the heat transfer properties of a mold with 0.25-inch of steel between the internal cavity and its cooling water jacket and a DLC film, a simple heat transfer calculation shows that the DLC layer will conduct heat 250 times faster than the thick tool steel.
Thickness. The typical DLC coating thickness for mold applications is between 3 and 4 microns (0.00012 and 0.00016 inch), which is extremely thin and allows it to be added to molds that may not have been specifically designed to include a coating.
The thickness of the coating is based on the substrate material and surface parameter optimizations. For example, different thicknesses may benefit different applications based on the substrate and desired combination of hardness, toughness and thermal transfer properties. Because the coatings are typically much thinner than 5 microns, they are much lower than most mold machining tolerances. This permits integration of the coating on many molds that are properly built to drawing tolerances.
Temperature. DLCs also have a low-deposition temperature that helps to eliminate tempering and thermal-related distortions. With common mold materials such as D2 steel, the temperature is less than 300ºF, and with QC-10 aluminum it is less than 250ºF. Inadvertent tempering of the material while applying a DLC changes the hardness of the substrate. By maintaining much lower deposition temperatures, the potential for tempering is dramatically reduced. The higher temperature DLC process makes thin film coating of theses molds impossible due to the changes in the substrate material properties.
Depositing DLCs
There are multiple commercially viable methods for depositing these films, all of which use a vacuum chamber setup. With this approach, molds and mold components are thoroughly cleaned prior to loading into a chamber to remove oils, scaling, oxidation and other surface contaminants. They are then loaded into the deposition system, which is pumped to a very low pressure (1×10-6 Torr or less). After additional surface cleaning (etching) in the chamber, the coatings are built through the bombardment of carbon atoms and other elements at a high enough ion energy level to create chemical bonding between the introduced atoms and the surface, as well as to each other.
While a small amount of methane is a byproduct of deposition, the actual coating is inert and not an environmental hazard. Through either a wet chemistry process or plasma etching, DLC coatings can be completely removed and then recoated; modified through machining or welding, and recoated; or chemically etched and recoated, as required.
Every DLC coating consists of an amorphous (non-crystalline) mixture of diamond and graphitic-phased carbon. The inclusion of elemental dopants in many of these recipes can help control film stress, provide reduced surface energy, enhance wear characteristics or create an advantageous tailored characteristic. The elemental dopants are non-carbon additives introduced at very low levels to produce specific material characteristics. For example, adding a metal dopant to enhance oleophilic (affinity for oil) behavior.
In the case of aluminum, carbides are not natively formed on the surface, so carbon ion implantation is required to establish anchor points for the coating. Based on this approach, there are many DLC recipes for producing the desired coating morphology (the microstructure of the metal surface) and tribological (friction, wear, lubrication and interface parameters of a material) performance parameters.
This well-balanced combination of properties allows DLC coatings to increase mold life, eliminate part sticking and reduce cycle time.
Related Content
Machine Hammer Peening Automates Mold Polishing
A polishing automation solution eliminates hand work, accelerates milling operations and controls surface geometries.
Read MoreHow to Use Thermal Management to Improve Mold Cooling
A review of common mold cooling issues and possible solutions, including 3D printing applications.
Read MorePredictive Manufacturing Moves Mold Builder into Advanced Medical Component Manufacturing
From a hot rod hobby, medical molds and shop performance to technology extremes, key relationships and a growth strategy, it’s obvious details matter at Eden Tool.
Read MoreSurface Finish: Understanding Mold Surface Lingo
The correlation between the units of measure used to define mold surfaces is a commonly raised question. This article will lay these units of measure side by side in a conversion format so that companies can confidently understand with what they are dealing.
Read MoreRead Next
How to Use Continuing Education to Remain Competitive in Moldmaking
Continued training helps moldmakers make tooling decisions and properly use the latest cutting tool to efficiently machine high-quality molds.
Read MoreReasons to Use Fiber Lasers for Mold Cleaning
Fiber lasers offer a simplicity, speed, control and portability, minimizing mold cleaning risks.
Read MoreAre You a Moldmaker Considering 3D Printing? Consider the 3D Printing Workshop at NPE2024
Presentations will cover 3D printing for mold tooling, material innovation, product development, bridge production and full-scale, high-volume additive manufacturing.
Read More















_300x250 4.png;maxWidth=300;quality=90)



.jpg;maxWidth=300;quality=90)